Pharmacokinetic & Bioequivalence Studies1,2
Mean Methylphenidate Plasma Concentrations Over Time
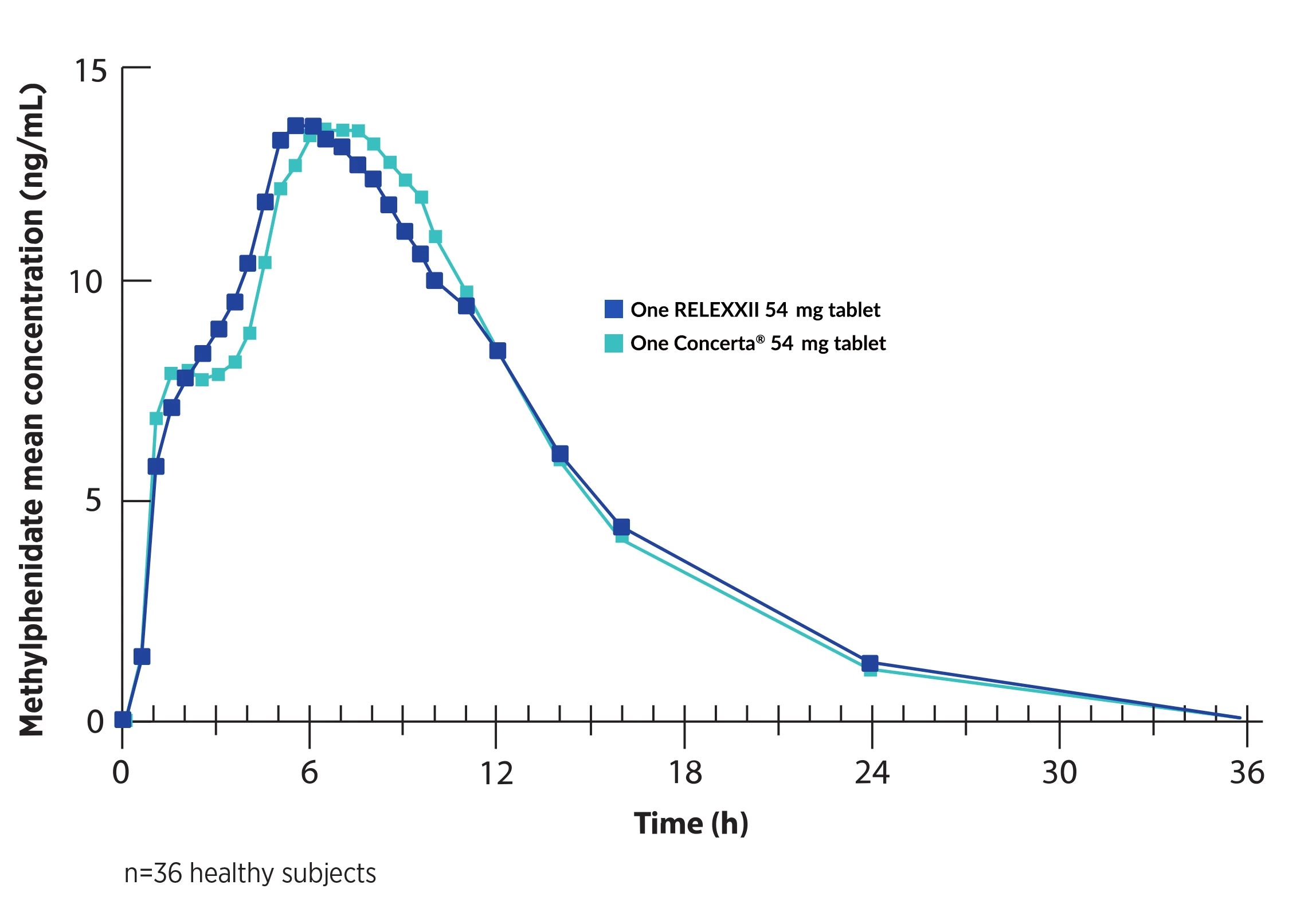
RELEXXII 54 mg
One RELEXXII 54 mg tablet has demonstrated bioequivalence to one Concerta® 54 mg tablet, including in the critical 7–12 hour post-dose window.1-3
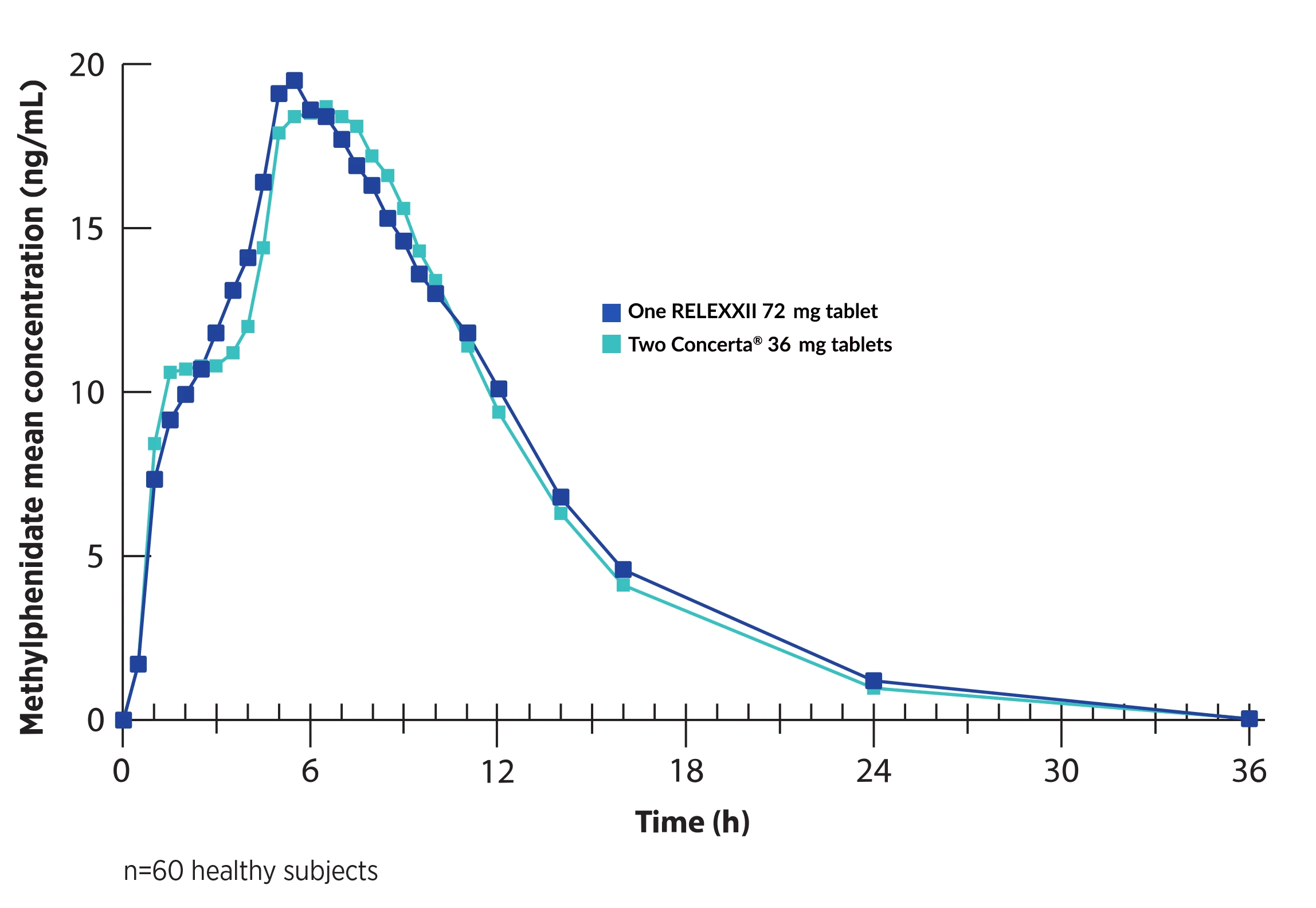
RELEXXII 72 mg
One RELEXXII 72 mg tablet has demonstrated bioequivalence to two Concerta® 36 mg tablets, including in the critical 7–12 hour post-dose window.1-3
Meets FDA Bioequivalent Criteria (Cmax, AUC, Tmax) to Concerta®.
View the Open-Access Clinical Publication
Pharmacokinetics and Bioequivalence of a Novel Extended-Release Formulation of Methylphenidate Hydrochloride for Attention-Deficit/Hyperactivity Disorder3
RELEXXII Offers Patients Reliable, All-Day ADHD Symptom Control With Just One Extended-Release Tablet
Symptom Control Throughout the Day
Help Improve Classroom Attention and Behavior with RELEXXII
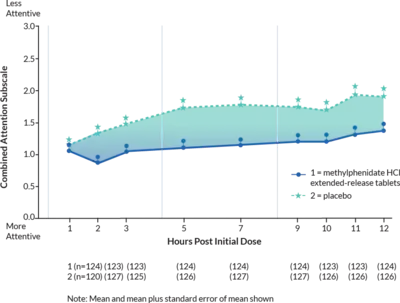
Combined results from two studies demonstrated statistically significant improvements in attention and behavior that were maintained through 12 hours after dosing.1
In Studies 1 and 2, symptoms of ADHD were evaluated by laboratory school teachers using the Swanson, Kotkin, Agler, M-Fynn, and Pelham (SKAMP) laboratory school rating scale.1
Study Design: Three doubleblind, active- and placebo controlled studies were conducted in 416 pediatric patients 6 to 12 years. The controlled studies compared methylphenidate hydrochloride extended-release tablets given once daily (18 mg, 36 mg, or 54 mg), methylphenidate given three times daily over 12 hours (15 mg, 30 mg, or 45 mg total daily dose), and placebo in two single-center, 3-week crossover studies (Studies 1 and 2).1
Laboratory School Teacher SKAMP Ratings:
Mean (SEM) of
Combined Attention (Studies 1 and 2)1
Based on FDA bioequivalence to another formulation of methylphenidate hydrochloride extended-release tablets. Studies not performed on RELEXXII.
Graph adapted from the PI.
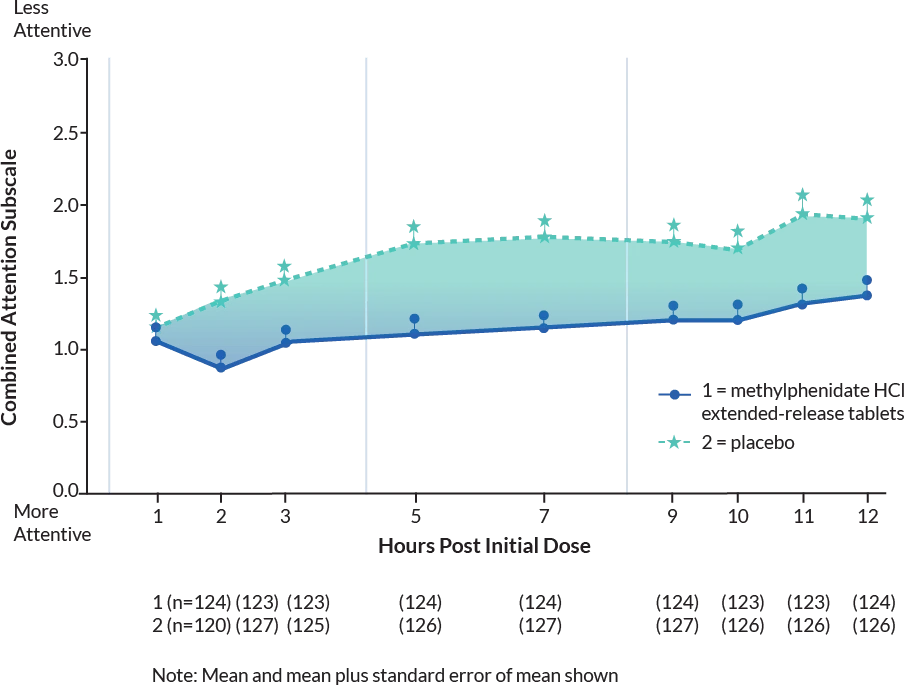
Laboratory School Teacher SKAMP Ratings:
Mean (SEM) of
Combined Attention (Studies 1 and 2)1
Concerta® is a registered trademark of ALZA Corporation.
References: 1. RELEXXII package insert. Vertical Pharmaceuticals, LLC. 2. Data on file, Osmotica Pharmaceuticals. 3. Childress AC, et al. Clin Pharmacol Drug Dev. 2025;0(0):1-7.
*Co-pay assistance is applicable to prescription coverage for eligible commercially insured patients. Offer void where prohibited. No income restrictions apply. Other restrictions may apply. Offer effective 04/2024. Alora Pharmaceuticals, LLC reserves the right to rescind, revoke, or amend this offer without notice. For full terms and conditions, click here.
All individuals depicted are models used for illustrative purposes only.



